|
12/21/2023 0 Comments Chlorines atomic radius
The general trend is that atomic sizes increase as one moves downwards in the Periodic Table of the Elements, as electrons fill outer electron shells. Atomic RadiiĪtomic radii represent the sizes of isolated, electrically-neutral atoms, unaffected by bonding topologies. In the following article we'll explore a number of different sets of distinct atomic radius sizes, and later we'll see how you can make use of these "preset" values with CrystalMaker. Take the humble carbon atom as an example: in most organic molecules a covalently-bonded carbon atom is around 1.5 Ångstroms in diameter (1 Ångstrom unit = 0.1 nanometres = 10 -10 metres) but the same atom in an ionic crystal appears much smaller: around 0.6 Ångstroms. However, even for atoms of the same type, atomic radii can differ, depending on the oxidation state, the type of bonding and - especially important in crystals - the local coordination environment. To a first approximation we can regard atoms as "hard spheres", with an outer radius defined by the outer electron orbitals. Donec aliquet.How big is an atom? A simple question maybe, but the answer is not at all straighforward. Fusce dui lectus, congue vel laoreet ac, dictum vitae odio. S ante, dapibus a molestie consequat, ultrices ac magna. Nam risus ante, dapibus a molestie consequat, ultrices ac magna. Nam lacinia pulvinar tortor nec facilisis. Lorem ipsum dolor sit amet, consectetur adipiscing elit. Nam risus ante, dapibus a molestie consequat, ultrices ac mag Risus ante, dapibus a molestie consequat, ultriĭonec aliquet. S a molestie consequat, ultrices ac magna. Nam risus ante, dapibus a molestie consequat Be sure to include forces and energy in your explanation.Įc facilisis. (4 points) Use your response to part e to explain why the sixth ionization energy for phosphorus is larger than the sixth ionization energy for chlorine. 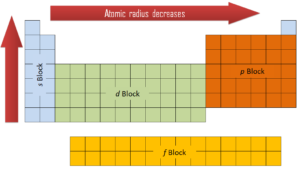
Use your electron configurations from part a to identify which orbital loses an electron corresponding to the sixth ionization energy for each element.į. 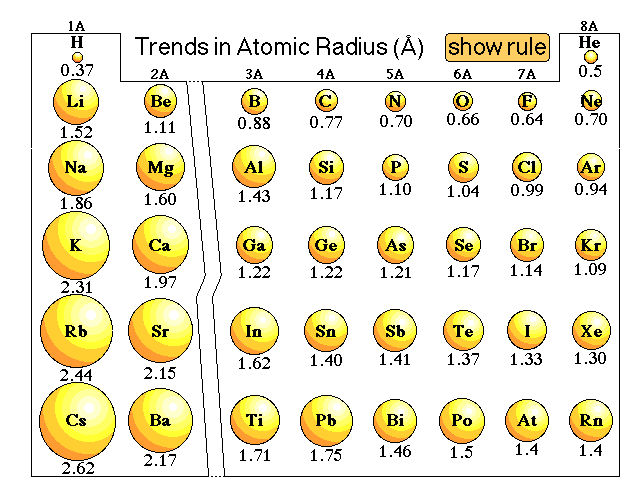
(2 points) The sixth ionization energy for phosphorus is 22,233 kJ/mol and the sixth ionization energy for chlorine is 9,458 kJ/mol. Be sure to include forces and energy in your explanation.Į. 
Use your evidence from parts a and b to explain why the first ionization energy of chlorine is larger than phosphorus. (3 points) The first ionization energy for phospohrus is 1012 kJ/mol and the first ionization energy for chlorine is 1251 kJ/mol. Be sure to include Coulomb's Law and forces in your explanation.ĭ. (3 points) Use your evidence from parts a and b to explain why the atomic radius of chlorine is smaller than the atomic radius of phosphorus. (2 points) Calculate the effective nuclear charge for P and Cl. (2 points) Write the full electron configuration for P and Cl.ī. The atomic radius of phosphorus is 195 pm and the atomic radius of chlorine is 175 pm.Ī.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
